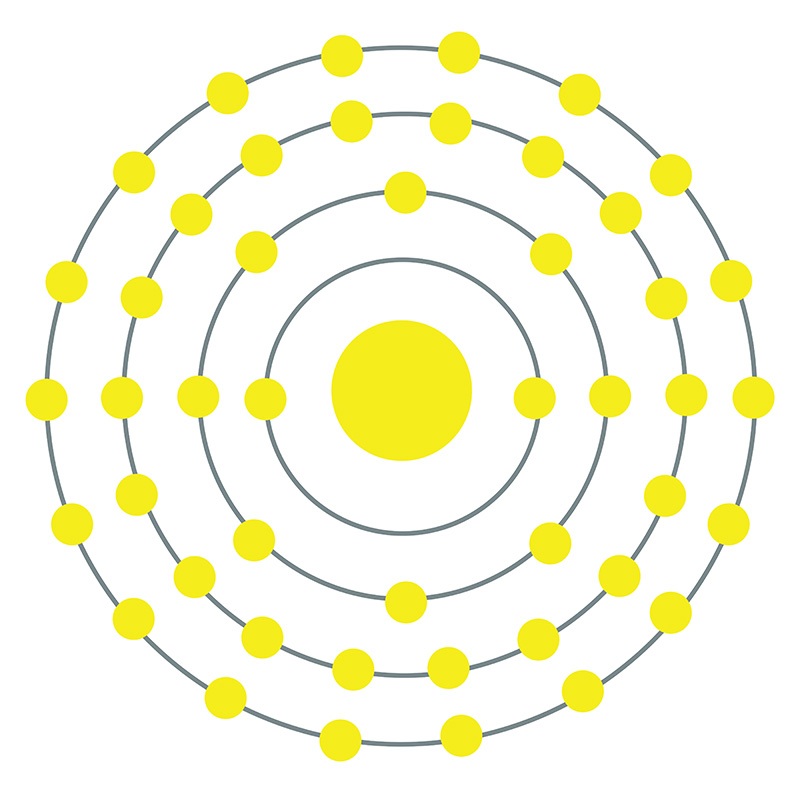
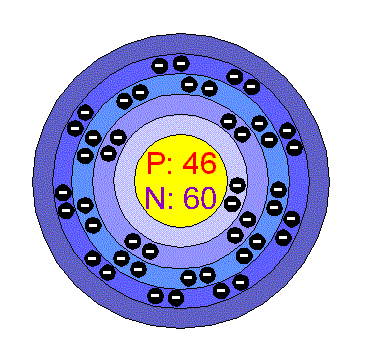
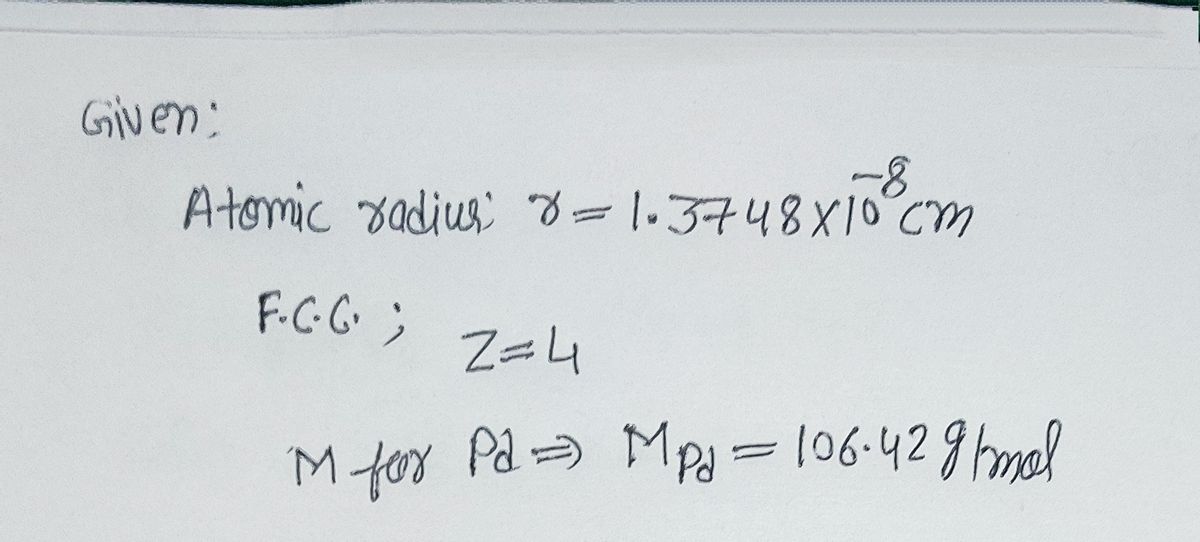HW2 answer 1 - 3.9 Calculate the radius of a palladium Pd atom given that Pd has an FCC crystal structure a density of 12.0 g/cm3 and an atomic | Course Hero

The atomic radius of palladium is 1.375 A. The unit cell of palladium is a face-centred cube. Ca... - YouTube