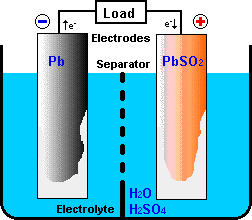
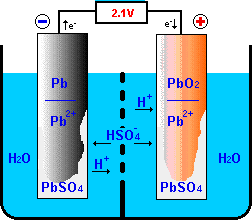
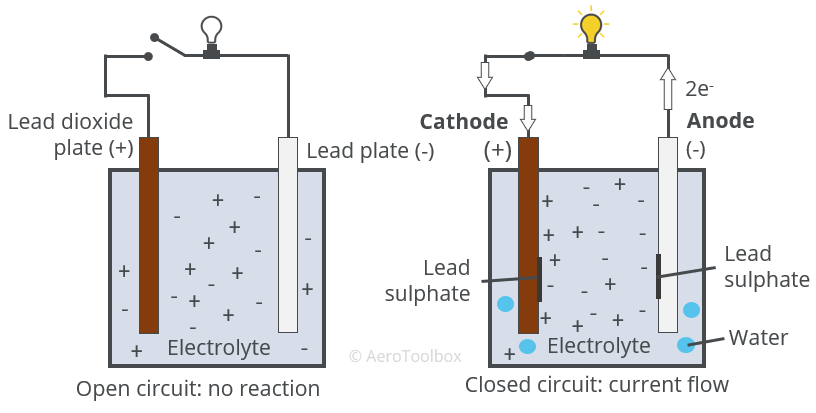
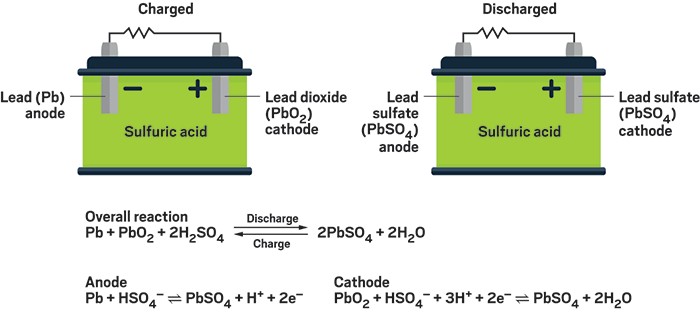
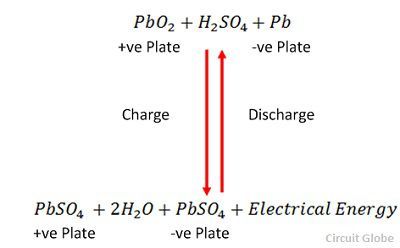
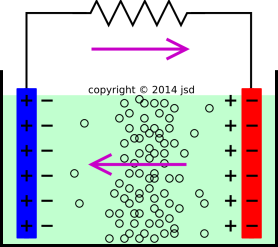
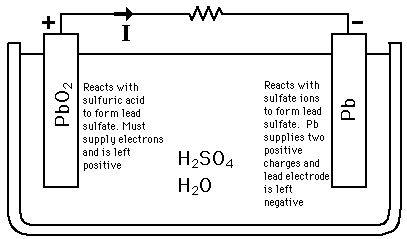
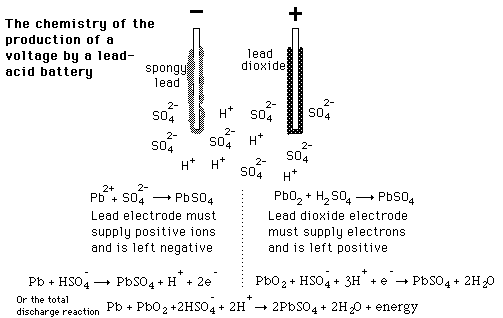The half reactions that occur in a lead acid battery are: `PbSO_(3)(s)+2e^(-)toPb(s)+SO_(4) - YouTube

Question Video: Identifying the Reaction That Occurs at the Positive Plate of a Lead-Acid Accumulator Battery During the Discharge Phase | Nagwa